Coal GCV Calculation From
Ultimate Analysis
The
main chemical elements in coal (apart from associated mineral matter) are C, O,
H, N and S. The chemical analysis is very important to calculate material
balance accurately and calorific value of coal. For the ultimate analysis C, H,
S and N are determined by chemical analysis and expressed on a moisture free
basis.
The
C, H, S and N produce heat on burning in presence of oxygen. The maximum heat
can only be achieved on complete combustion.
The
heat generated by element on burning with oxygen is as follows
|
C |
+ |
O2 |
= |
CO2 |
+ |
8080 kcal/Kg |
|
2C |
+ |
O2 |
= |
2CO |
+ |
2430 kcal/Kg |
|
H2 |
+ |
1/2O2 |
= |
H2O |
+ |
34500 Kcal/Kg |
|
S |
+ |
O2 |
= |
SO2 |
+ |
2020 Kcal/Kg |
Maximum
heat is generated on burning is of Hydrogen. On burning of hydrogen it also forms
water which convert to steam by absorbing latent heat from reaction. Now the
amount of hydrogen involves for the formation of water will be
|
H2 |
+ |
1/2O2 |
= |
H2O |
+ |
34500 Kcal/Kg |
|
1X2 |
+ |
0.5X16X2 |
= |
(1X2)+16 |
|
|
|
2 |
+ |
16 |
= |
18 |
|
|
|
2/2 |
+ |
16/2 |
= |
18/2 |
|
|
|
1 |
+ |
8 |
= |
9 |
|
|
From
the above equation it is found that 1 part of Hydrogen reacts with 8 part of
Oxygen to form 9 part of water. So now it can be concluded that ![]() O part of hydrogen
will form water (steam) on combustion. The amount of heat producing hydrogen in
coal will be
O part of hydrogen
will form water (steam) on combustion. The amount of heat producing hydrogen in
coal will be
![]() O
O
Now GCV of coal will be
calculated by adding all heat producing elements, which is expressed as Dulong’s Formula
Calorific Value = 8080XC + 34500X[![]() O]
+ 2020XS
O]
+ 2020XS
Case Study:
On ultimate analysis of a
coal sample following data was found,
|
C
= 30% |
O
= 3% |
H
= 2% |
S
= 0.5% |
As per Dulong’s Formula
Calorific Value = 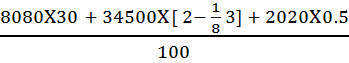 = 2995.725 Kcal
= 2995.725 Kcal